In February 2026, the biopharmaceutical spin-off of Google DeepMind, Isomorphic Labs, unveiled a groundbreaking artificial intelligence (AI) model designed to revolutionize drug discovery. Named IsoDDE, this new AI system marks a significant advancement in the field, particularly in predicting how proteins interact with potential therapeutic molecules. However, unlike DeepMind’s earlier AlphaFold models, which were openly shared with the scientific community, IsoDDE remains proprietary, sparking both admiration and frustration among researchers striving to replicate its success.
Nearly two years prior, DeepMind had released AlphaFold 3, an updated AI tailored for drug discovery. This version built upon the Nobel Prize-winning AlphaFold 2 by not only predicting protein structures but also modeling how proteins interact with other molecules—a critical step in understanding potential drug efficacy. Now, Isomorphic Labs claims to have surpassed these capabilities with IsoDDE, as detailed in a comprehensive 27-page technical report published on February 10, 2026.
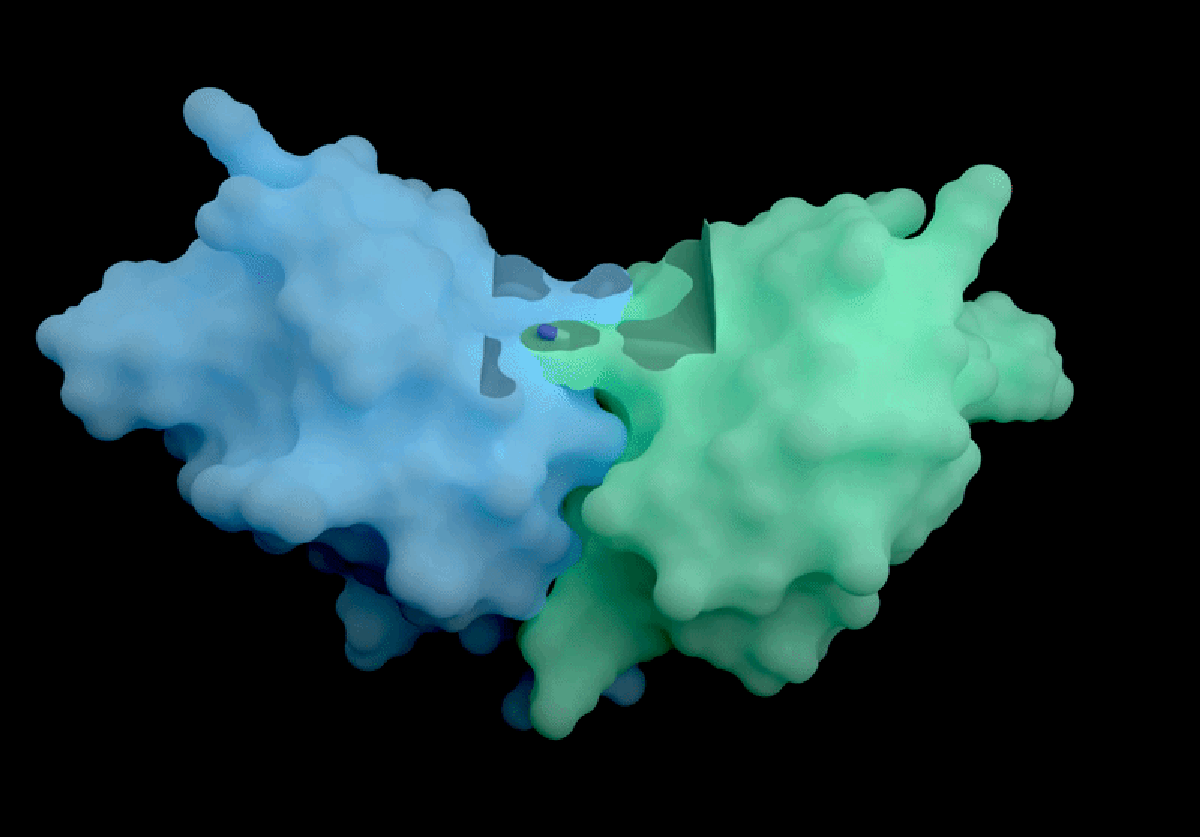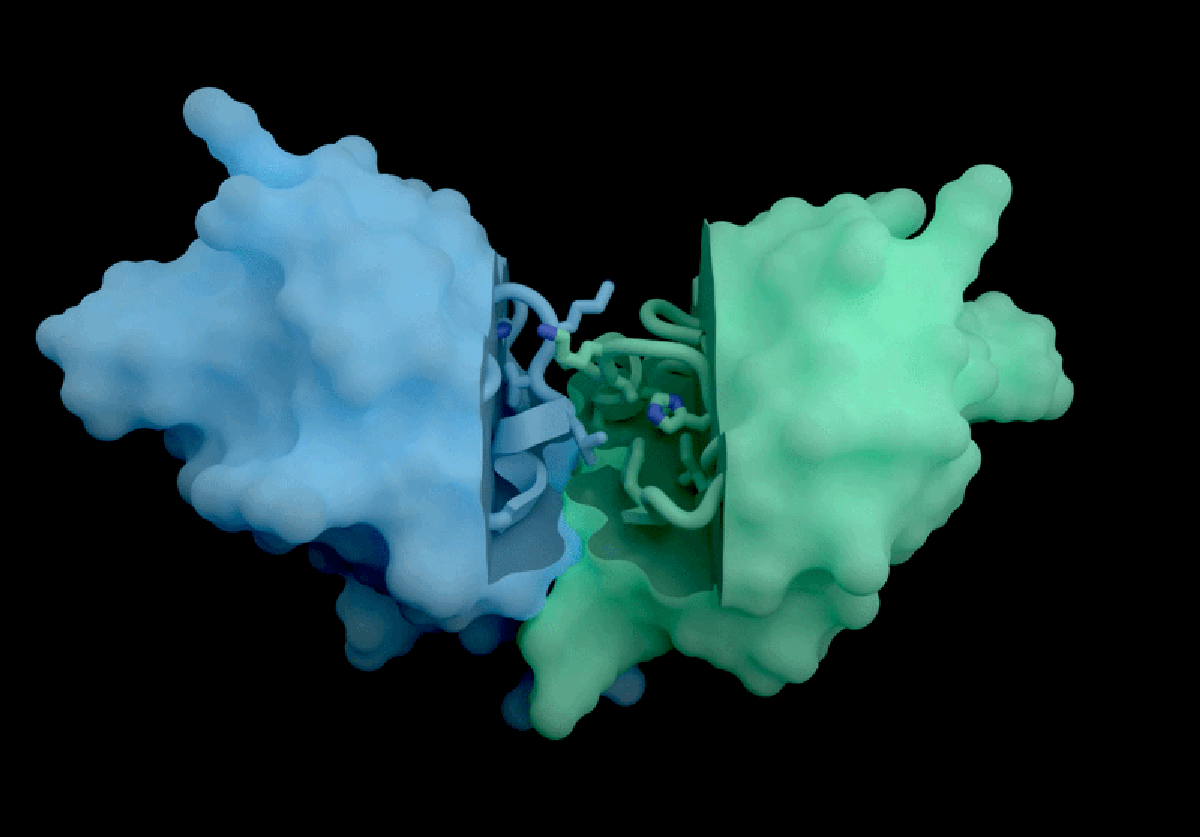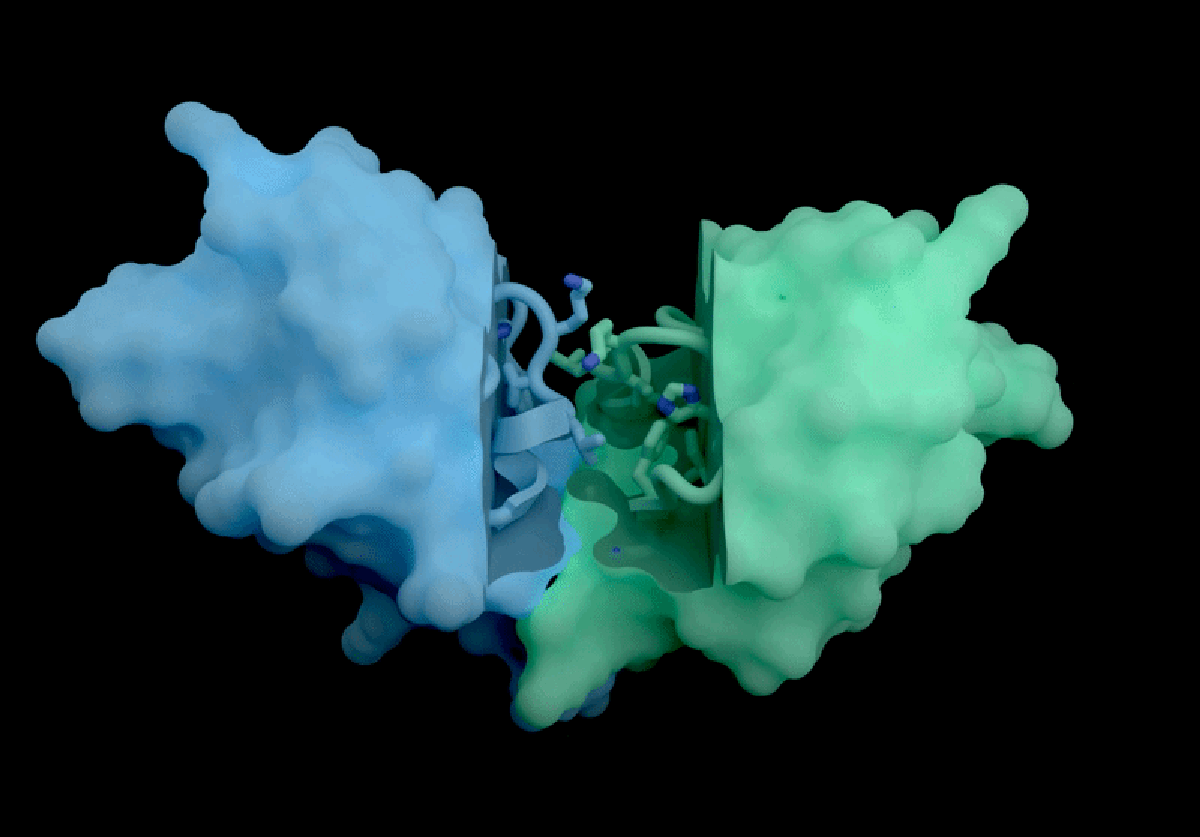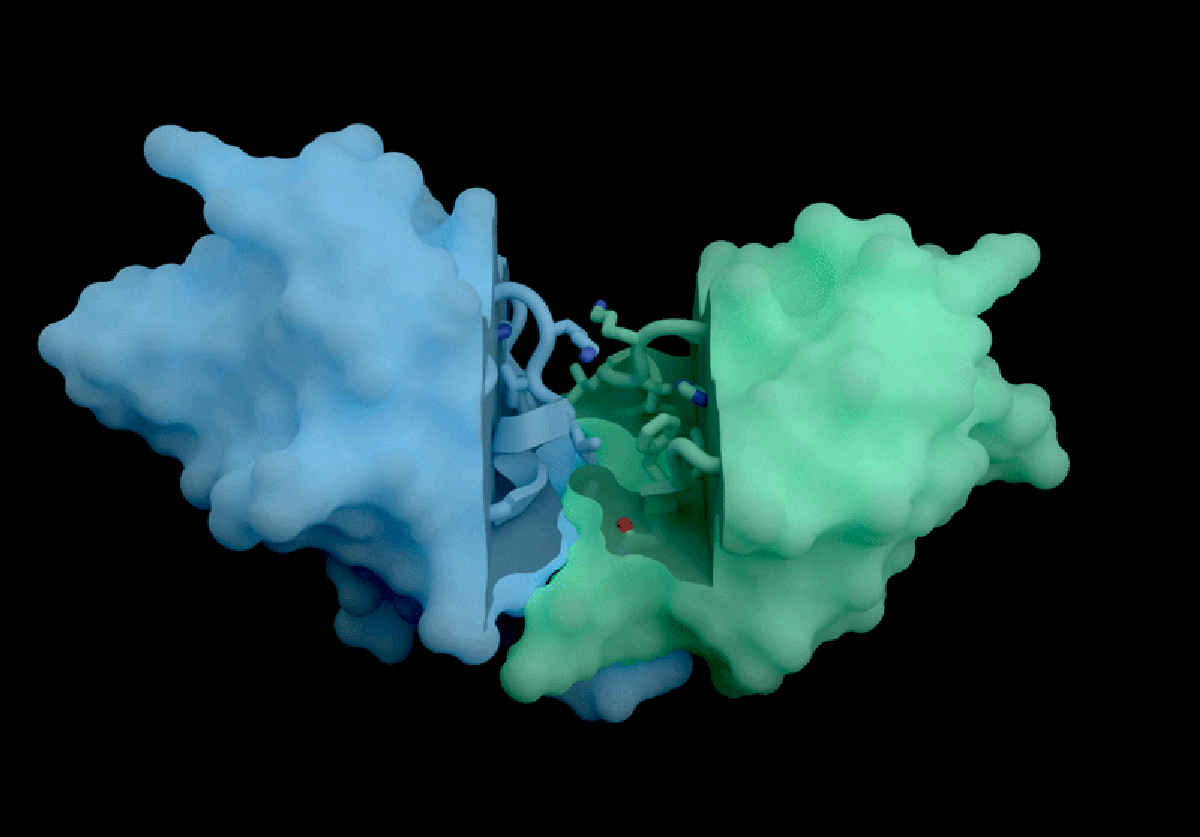
IsoDDE’s key strengths include highly accurate predictions of protein-ligand interactions, including binding affinity, which measures how strongly a potential drug molecule attaches to a target protein. Binding affinity is a fundamental property for developing effective therapeutics but is traditionally challenging to estimate accurately without computationally expensive physics-based simulations. According to Isomorphic Labs, IsoDDE outperforms not only these traditional methods but also other AI models inspired by AlphaFold 3, such as Boltz-2, an open-source tool developed by researchers at the Massachusetts Institute of Technology (MIT).
Boltz-2, released last year, demonstrated impressive capabilities in predicting binding affinities and has been seen as a leading open-source competitor in the drug discovery AI space. However, Isomorphic Labs’ new model reportedly exceeds Boltz-2’s performance, particularly in predicting interactions involving antibodies—proteins that underpin many of the most lucrative therapies worldwide. This ability to accurately model antibody-target interactions could accelerate the development of new treatments worth billions of pounds annually.
Experts in the field are highly impressed by IsoDDE’s reported performance, especially its capacity to predict interactions involving molecules that differ significantly from the data it was trained on. Mohammed AlQuraishi, a computational biologist at Columbia University who works on open-source AlphaFold variants, described the model as “a major advance, on the scale of an AlphaFold 4,” referencing an as-yet-unreleased future generation of DeepMind’s technology. He emphasizes, however, that the lack of detailed information about IsoDDE’s inner workings leaves scientists “guessing” about how to replicate or build upon these results.
Isomorphic Labs’ president, Max Jaderberg, acknowledges that the models behind IsoDDE are “profoundly different” from other efforts but declines to disclose the “secret sauce” that enables its performance. He attributes IsoDDE’s success to a combination of computational power, data, and novel algorithms—a common refrain in AI development—but remains tight-lipped about specifics. Jaderberg hopes that the company’s technical report will inspire other research teams to accelerate their work in drug-discovery AI, even without access to IsoDDE’s proprietary details.
The proprietary nature of IsoDDE has drawn criticism and curiosity from the scientific community. Whereas DeepMind’s earlier AlphaFold models were made openly available and thoroughly documented, facilitating widespread adoption and further research, IsoDDE’s closed approach limits transparency and broader validation. Diego del Alamo, a computational structural biologist at Takeda Pharmaceuticals, noted on social media that the report followed efforts to partner with industry and potentially access private structural data, leaving questions about how much exclusive data contributed to IsoDDE’s capabilities.
Isomorphic Labs is actively leveraging IsoDDE’s potential through partnerships with major pharmaceutical companies, including Johnson & Johnson, Eli Lilly, and Novartis. These collaborations could be worth billions of pounds, reflecting the enormous commercial interest in AI-driven drug discovery. Additionally, the company maintains its own drug development pipeline, with clinical trials anticipated in the near future. Jaderberg revealed that different versions of IsoDDE have been developed to meet the needs of various partners, incorporating diverse data sources beyond those described in the public report.
Michael Schaarschmidt, Isomorphic’s director of machine learning, highlighted the company’s comprehensive data strategy, which combines publicly available datasets, synthetic training data generated by the AI itself, and licensed proprietary data. This multifaceted approach affords IsoDDE access to a rich variety